Stanley Hazen Laboratory
-
Stanley Hazen Laboratory
- Principal Investigator
- Research
- Our Team
- Publications
- Careers
- Research News
Research
A long-term goal of my laboratory is to identify novel pathways that contribute to cardiovascular and metabolic diseases. I have several major areas of focus. One centers on the role of gut microbes as contributors to heart and metabolic (e.g., kidney, diabetes) diseases. Another area focuses on the recent discovery of sugar alcohols like erythritol and xylitol as contributors to cardiovascular disease. Yet another program focuses on how inflammation pathways are mechanistically linked to cardiovascular disease. All research programs span from bench to bedside, and involve both large-scale clinical studies and mechanistic (cellular, animal model) studies. Emphasis is focused on development of new therapeutics to target pathways that contribute to cardiovascular and metabolic diseases.
Biography
Stanley Hazen, MD, PhD, received clinical training in Internal Medicine and subspecialty training in Diabetes, Endocrinology and Metabolism from Barnes/Jewish Hospital, St. Louis, MO, and a PhD in Biophysical Chemistry and Molecular Biology from Washington University School of Medicine, St. Louis, MO. He holds multiple leadership positions at the Cleveland Clinic including chair, Department of Cardiovascular & Metabolic Sciences, Cleveland Clinic Research; co-Section head, Preventive Cardiology & Rehabilitation, Heart, Vascular & Thoracic Institute; and Director, Center for Microbiome & Human Health. Dr. Hazen sees patients within the Preventive Cardiology Clinic, specializing in preventive cardiovascular medicine care of patients including treatment of hyperlipidemia, hypertension, obesity and diabetes. He also sees patients within the Cardiovascular Rehabilitation program. His research interests include studies on mechanisms that contribute to the development of cardiovascular disease, with emphasis on understanding the role of the gut microbiome, inflammation and metabolism in the pathogenesis of atherosclerosis and other inflammatory diseases.
A renowned physician scientist, Dr. Hazen has made pioneering discoveries in new understandings of mechanisms contributing to cardiovascular and metabolic disease research. He is credited with numerous seminal discoveries linking gut microbial pathways to cardiovascular disease pathogenesis, as well as enumerating the role of myeloperoxidase and other inflammatory and oxidative pathways in cardiovascular disease. Dr. Hazen is among the top 0.1% cited researchers in the world with over 180,000 citations. He has published over 540 peer-reviewed articles in top-tier basic and clinical journals alike in the fields of atherosclerosis, thrombosis, lipoprotein metabolism, gut microbiome, heart failure, vascular biology, and other topics related to preventive cardiovascular medicine. His research in multiple areas has impacted clinical practice, and lays the foundation for FDA- and EU-cleared diagnostic tests for cardiovascular disease risk assessment in use worldwide. He is listed as inventor on over 100 patents, and his research has helped to spawn pharmaceutical development of cardiovascular disease drugs in clinical trials.
Dr. Hazen has received numerous awards including being the inaugural recipient of the Top 10 Clinical Discovery of the Year (2011) Award, Clinical Research Forum; the American Heart Association/American Stroke Association “top 10 advance in heart disease and stroke science” award (2013); and the American Heart Association Distinguished Scientist Award (2017). Dr Hazen has been honored with election of membership into honorary societies in both science and clinical arenas alike, including the American Society for Clinical Investigation (ASCI) and the American Association of Physicians (AAP). Dr. Hazen is an elected fellow of the American Association for the Advancement of Science (AAAS). Dr. Hazen also is an elected member of the National Academy of Medicine, USA.
Education & Professional Highlights
Education & Fellowships
Fellowship - Barnes-Jewish Hospital
Endocrinology
St. Louis, MO USA
1996
Residency - Barnes-Jewish Hospital
Internal Medicine
St. Louis, MO USA
1994
Medical Education (MD) - Washington University School of Medicine
St. Louis, MO USA
1992
Graduate Education (PhD) - Washington University School of Medicine
St. Louis, MO USA
1992
Undergraduate - Washington University
St. Louis, MO USA
1985
Professional Highlights
- Chair, Gordon Research Conference on Oxygen Radicals
- Chair, Gordon Research Conference on Atherosclerosis
- Jeffrey M Hoeg Arteriosclerosis, Thrombosis and Vascular Biology Award for Basic Science and Clinical Research
- Elected member, Fellow of American Heart Association Scientific Council for Arteriosclerosis
- Elected member, American Society for Clinical Investigation (ASCI)
- Elected member, Association of American Physicians (AAP)
- Elected fellow, American Association for the Advancement of Science (AAAS).
- Elected member, The National Academy of Medicine, USA.
Awards & Honors
- Howard Hughes Medical Institute Research Fellowship for Physicians Award, 1995-1997
- AAP, ASCI, and AFMR Certificate of Achievement Award, 1997
- Marilyn Hansen American Thoracic Society Award, 1997
- Junior Faculty Award, American Federation of Medical Research, 1999
- Gill Heart Institute Physician Scientist Award, University of Kentucky, Lexington Ky., 2001
- Election, American Society for Clinical Investigation, 2003
- John J. Ferchill Award for Innovation, Cleveland Clinic, 2004
- Nomination, Howard Hughes Medical Institute Investigator Award, 2004
- Vice-Chair, Gordon Research Conference on Oxygen Radicals, 2006
- Jeffrey M Hoeg Arteriosclerosis, Thrombosis and Vascular, 2007
Innovations & Patents
- Patent Title: Diagnostic Methods for Asthma; U.S. Serial Number 09/253,380
- Patent Title: Myeloperoxidase, a Risk Indicator for Cardiovascular Disease; U.S., Serial Number 10/039,753
- Patent Title: Treating Inflammation and Associated Complications; Patent Reference Number 09531-030P01 (Pending)
- Patent Title: Methods of Identifying Subjects at Increased Risk for Cardiovascular Disease; U.S. Serial Number 60/259,340 (Pending)
- Patent Title: Monitoring Anti-inflammatory and Anti-oxidant Actions of Therapies; U.S. Serial Number (Pending)
- Patent Title: Diagnostic Method for Identifying Subjects at Risk for Atherosclerosis and its Complications; U.S. Serial Number (Pending)
Memberships
- American Society for Clinical Investigation
- American Association for the Advancement of Science
- American Heart Association Scientific Council for Arteriosclerosis
- American Society for Investigative Pathology
- American College of Cardiology
Research
Overview
A long-term goal of my laboratory is to understand mechanisms through which inflammation contributes to diseases like atherosclerosis and chronic kidney disease. Several major research programs are currently under investigation. One research program focuses on the role of the gut microbiome in cardiovascular, kidney and metabolic diseases. A second area focuses on development of novel therapeutic approaches to the treatment of residual cardiovascular disease risks, kidney and metabolic disorders.
All research projects rely heavily on chemical and analytical methods to identify specific reactions/products, their mechanisms of formation, and their use as probes to elaborate pathways responsible for disease. Research efforts in each program span from bench to bedside, including basic/genetic, cellular, animal model, and human clinical investigations. A major emphasis is the combined use of large-scale clinical studies to define hypotheses, and development of therapeutic interventions for the treatment of disorders.
Experimental Data
Experimental data and computational models for downloading
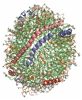
1) The refined structure of nascent HDL reveals a key functional domain for particle maturation and dysfunction
Wu, Z.; Wagner, M. A.; Zheng, L.; Parks, J. S.; Shy II, J. M.; Smith, J. D.; Gogonea, V.; Hazen, S. L. Nat. Struct. Mol. Biol. 14, 861-8 (2007).
Computational model:
 |
a) All-atom molecular model of the Solar Flares model of nascent HDL. Available at http://mi.caspur.it/PMDB/main.php (accession code PM0074956). |
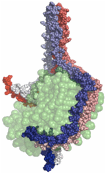
2) The double super helix model of high density lipoprotein
Wu, Z.; Gogonea, V.; Lee, X.; Wagner, M. A.; Li, X.-M.; Huang, Y.; Arundhati, U.; May, R. P.; Haertlein, M.; Moulin, M.; Gutsche, I.; Zaccai, G.; DiDonato, J.; Hazen, S. L. J. Biol. Chem. 284, 36605-19 (2009).
Experimental data and computational models:
|
a) Small angle neutron scattering intensities for the protein component of nascent HDL (12 % D2O, collected at the Institute Laue-Langevin, Grenoble, France). |
|
|
b) The solution low-resolution structure of the protein component of nascent HDL obtained by small angle neutron scattering with contrast variation (12 % D2O, ILL data). |
|
|
c) Small angle neutron scattering intensities for the lipid component of nascent HDL (42 % D2O, collected at the Institute Laue-Langevin, Grenoble, France). |
|
 |
d) The solution low-resolution structure of the lipid component of nascent HDL obtained by small angle neutron scattering with contrast variation (42 % D2O, ILL data). |
 |
e) The Double Super Helix model of nascent HDL. Available at www.rcsb.org (accession code 3K2S). |
3) Congruency between biophysical data from multiple platforms and molecular dynamics simulation of the double super helix model of nascent high-density lipoprotein
Gogonea, V.; Wu, Z.; Lee, X.; Pipich, V.; Li, X.-M., Ioffe, I. A.; DiDonato, J.; Hazen, S. L. Biochemistry, 49, 7323-43 (2010).
Experimental and computational models, and data calculated from simulation trajectory:
4) The low resolution structure of ApoA1 in spherical high density lipoprotein revealed by small angle neutron scattering
Wu, Z.; Gogonea, V.; Lee, X.; May, R.P.; Pipich, V.; Wagner, M.A.; Undurti, A.; Tallant, T. C.; Baleanu-Gogonea, C.; Charlton, F.; Ioffe, I. A.; DiDonato, J.A.; Rye, K.-A.; Hazen, S. L. J. Biol. Chem., 286, 12495-508 (2011).
Low resolution structures:
|
a) Small angle neutron scattering intensities for the protein component of spherical HDL (12 % D2O). |
|
 |
|
|
c) Small angle neutron scattering intensities for the lipid component of spherical HDL (42 % D2O). |
|
 |
5) The low resolution structure of nascent high density lipoprotein reconstituted with DMPC with and without cholesterol reveals a mechanism for particle expansion
Gogonea, V.; Gerstenecker, G. S.; Wu, Z.; Lee, X.; Topbas, C.; Wagner, M. A.; Tallant, T. C.; Smith J. D.; Callow, P.; Pipich, V.; Malet, H.; Schoehn, G.; DiDonato, J. A.; Hazen, S. L. J. Lipid Res., in print.
Low resolution structures and computational model:
Our Team
Selected Publications
View publications for Stanley Hazen, MD, PhD
(Disclaimer: This search is powered by PubMed, a service of the U.S. National Library of Medicine. PubMed is a third-party website with no affiliation with Cleveland Clinic.)
- Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WHW, DiDonato JA, Lusis AJ, Hazen SL. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature.(2011) 472(7341):57-63. PMCID: PMC3086762
- Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL. Intestinal Microbial Metabolism of Phosphatidylcholine and Cardiovascular Risk. New England Journal of Medicine. (2013) 368(17):1575-84. PMCID: PMC3701945
- Koeth RA, Wang Z, Levison BS, Buffa J, Org E, Sheehy B, Li H, Britt EB, Fu X, Wu Y, Smith JD, DiDonato JA, Chen J, Li H, Wu G, Lewis JD, Warrier M, Brown, JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, and Hazen SL. Intestinal microbiota metabolism of L-Carnitine, a nutrient in red meat, promotes atherosclerosis. Nature Medicine. (2013) 19(5): 576-85. PMCID: PMC36501111
- Huang Y, Didonato JA, Levison BS, Schmitt D, Li L, Wu Y, Buffa J, Kim T, Gerstenecker GS, Gu X, Kadiyala CS, Wang Z, Culley MK, Hazen JE, Didonato AJ, Fu X, Berisha SZ, Peng D, Nguyen TT, Liang S, Chuang CC, Cho L, Plow EF, Fox PL, Gogonea V, Tang WH, Parks JS, Fisher EA, Smith JD, Hazen SL. An abundant dysfunctional apolipoprotein A1 form in human atheroma. Nature Medicine. (2014) 20(2):193-203. PMCID: PMC3923163
- Wang Z, Roberts AB, Buffa JA, Levison BS, Zhu W, Org E, Gu X, Huang Y, Zamanian-Daryoush M, Culley MK, DiDonato AJ, Fu X, Hazen JE, Krajcik D, DiDonato JA, Lusis AJ, Hazen SL. Non-lethal Inhibition of Gut Microbial Trimethylamine Production for the Treatment of Atherosclerosis. Cell. 2015;163(7):1585-95. Epub 2015/12/22. doi: 10.1016/j.cell.2015.11.055. PubMed PMID: 26687352; PMCID: PMC4871610.
- Zhu W, Gregory JC, Org E, Buffa JA, Gupta N, Wang Z, Li L, Fu X, Wu Y, Mehrabian M, Sartor RB, McIntyre TM, Silverstein RL, Tang WHW, DiDonato JA, Brown JM, Lusis AJ, Hazen SL. Gut Microbial Metabolite TMAO Enhances Platelet Hyperreactivity and Thrombosis Risk. Cell. 2016;165(1):111-24. Epub 2016/03/15. doi: 10.1016/j.cell.2016.02.011. PubMed PMID: 26972052; PMCID: PMC4862743.
- Nemet I, Saha PP, Gupta N, Zhu W, Romano KA, Skye SM, Cajka T, Mohan ML, Li L, Wu Y, Funabashi M, Ramer-Tait AE, Naga Prasad SV, Fiehn O, Rey FE, Tang WHW, Fischbach MA, DiDonato JA, Hazen SL. A Cardiovascular Disease-Linked Gut Microbial Metabolite Acts via Adrenergic Receptors. Cell. 2020;180(5):862-77.e22. Epub 2020/03/07. doi: 10.1016/j.cell.2020.02.016. PubMed PMID: 32142679; PMCID: PMC7402401.
- Buffa JA, Romano KA, Copeland MF, Cody DB, Zhu W, Galvez R, Fu X, Ward K, Ferrell M, Dai HJ, Skye S, Hu P, Li L, Parlov M, McMillan A, Wei X, Nemet I, Koeth RA, Li XS, Wang Z, Sangwan N, Hajjar AM, Dwidar M, Weeks TL, Bergeron N, Krauss RM, Tang WHW, Rey FE, DiDonato JA, Gogonea V, Gerberick GF, Garcia-Garcia JC, Hazen SL. The microbial gbu gene cluster links cardiovascular disease risk associated with red meat consumption to microbiota L-carnitine catabolism. Nat Microbiol. 2022;7(1):73-86. Epub 2021/12/25. doi: 10.1038/s41564-021-01010-x. PubMed PMID: 34949826; PMCID: PMC8732312.
- Zhu Y, Dwidar M, Nemet I, Buffa JA, Sangwan N, Li XS, Anderson JT, Romano KA, Fu X, Funabashi M, Wang Z, Keranahalli P, Battle S, Tittle AN, Hajjar AM, Gogonea V, Fischbach MA, DiDonato JA, Hazen SL. Two distinct gut microbial pathways contribute to meta-organismal production of phenylacetylglutamine with links to cardiovascular disease. Cell Host Microbe. 2023;31(1):18-32.e9. Epub 20221221. doi: 10.1016/j.chom.2022.11.015. PubMed PMID: 36549300; PMCID: PMC9839529.
- Saha PP, Gogonea V, Sweet W, Mohan ML, Singh KD, Anderson JT, Mallela D, Witherow C, Kar N, Stenson K, Harford T, Fischbach MA, Brown JM, Karnik SS, Moravec CS, DiDonato JA, Naga Prasad SV, Hazen SL. Gut microbe-generated phenylacetylglutamine is an endogenous allosteric modulator of β2-adrenergic receptors. Nature Commun. 2024 Aug 6;15(1):6696. doi: 10.1038/s41467-024-50855-3. PMID: 39107277; PMCID: PMC11303761.
Careers
Training at Cleveland Clinic Research
Our education and training programs offer hands-on experience at one of the nationʼs top hospitals. Travel, publish in high impact journals and collaborate with investigators to solve real-world biomedical research questions.
Learn MoreResearch News

Recent cardiovascular research at Cleveland Clinic examined topics such as the gut microbiome connection to the heart, COVID-19’s impact on cardiac event risk and improving cardiac imaging.

UK Biobank analysis reveals risk of cardiac events stays elevated for years after diagnosis.

Study shows beta-2 adrenergic receptors can be regulated from a new site which allows healthy signals through, minimizing harmful side effects of beta-blocker medications.

The pathway serves as a strong biomarker for predicting risk for developing heart failure and could be a target for therapeutics.

Participants showed a higher potential for blood clotting after consuming a beverage sweetened with the sugar substitute erythritol compared to sugar.

The findings raise concerns about the safety of consuming sugar alcohols, especially for those at increased risk for cardiac disease.

Study outlines genetic links between 4PY and vascular inflammation, indicates need for future research.

Inching closer to precision heart care medicine, Cleveland Clinic researchers develop an “atlas” of the human microbiome pathways linked to cardiac and mortality risks.

The additive’s clinical association with cardiovascular risk, coupled with increased clotting in preclinical models, showcases the need for further safety studies.

Two distinct gut microbial enzyme pathways could be targeted in therapeutic development.